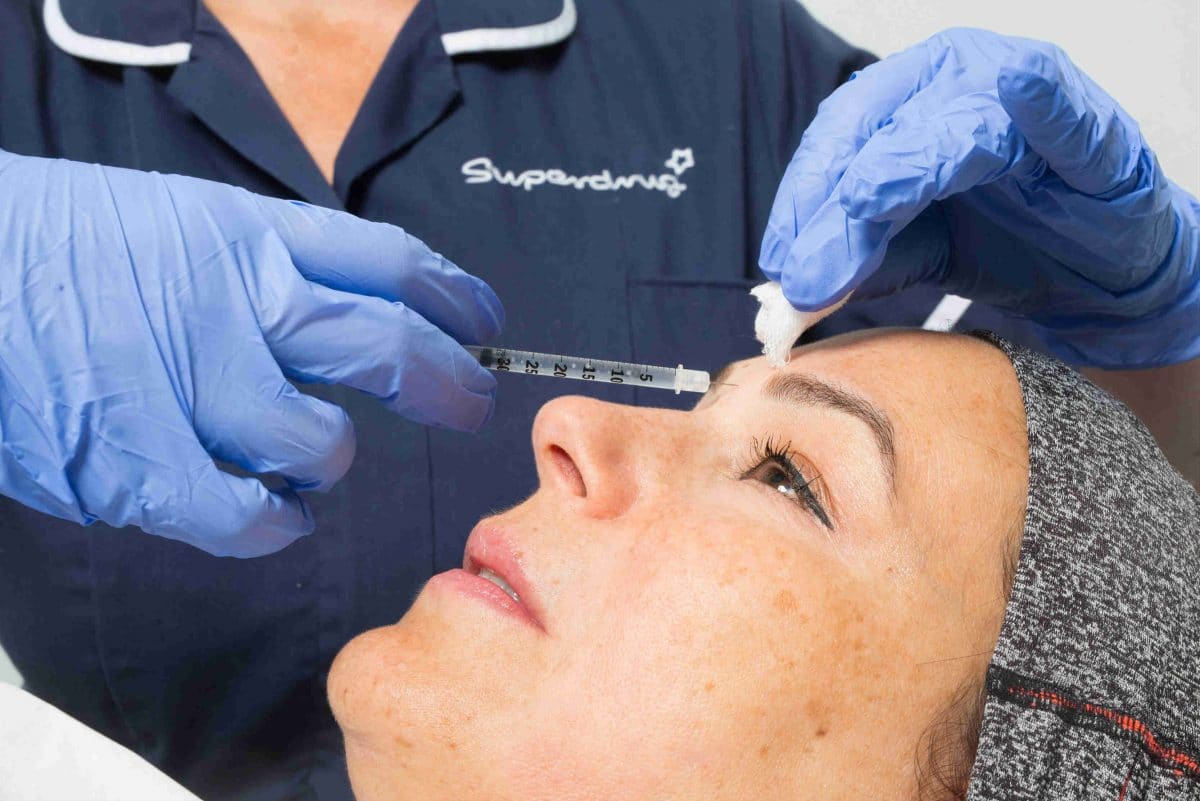
Superdrug has come under pressure from the NHS to screen customers before providing botox and fillers in order to protect vulnerable people.
The health and beauty retailer announced last month that it would offer the anti-wrinkle and skin rejuvenation treatments on the high street to customers over 25.
The Skin Renew Service, which has launched in Superdrug’s London Strand store store before being rolled out nationwide, is only available to customers who first book over the phone for a consultation appointment with a qualified nurse.
The procedures start at £99 and are carried out in a private consultation room.
NHS England national medical director Professor Stephen Powis has called for reassurances the service is “clinically sound and medically responsible”.
In a letter to Peter Macnab, chief executive of Superdrug’s parent company A.S. Watson, he said staff must be trained to check for people with body image disorder or “unrealistic expectations” about the procedures “in the interests of protecting potentially vulnerable members of the public”.
Professor Powis also said the treatments could pose a risk to patients.
“I understand that your company is set to offer cosmetic non-surgical procedures in your high street outlets,” he wrote.
“These interventions are invasive procedures and may be accompanied by serious risks.
“They should be offered only in situations where they are accompanied by a robust level of clinical governance, and they should be provided only by trained professionals with a full understanding of the implications and risks involved.”
Professor Powis requested details of the training and qualifications of the staff who will carry out the procedures and reassurances that clinicians and patients will be aware of possible complications.
Superdrug has also been asked to confirm it has adopted the professional standards for non-surgical cosmetic procedures.
“While I expect that all appropriate safeguards are in place for your service, you will know that the unchecked proliferation of providers offering cosmetic procedures introduces a risk to patient safety, unless strong and vigilant clinical risk assessments are established,” Professor Powis said.
A spokeswoman for Superdrug said: “Having reviewed the letter from Professor Stephen Powis we will be providing him with the full details on the qualifications of our practitioner and the processes we have in place to ensure the very highest standards of care and patient safety.
“We’re highly supportive of championing a more regulated service to further ensure patients’ safety in aesthetic treatments and would welcome the opportunity to work with the NHS England and other organisations to achieve this aim.”
Click here to sign up to Retail Gazette‘s free daily email newsletter